
Ocular in vivo Pharmacology

Disease Models for Ocular in vivo Pharmacology and Efficacy Testing
Pharmaron’s ocular in vivo pharmacology models provide a precise way to evaluate your candidate drug’s efficacy and pharmacology. By integrating clinically relevant preclinical models with advanced evaluation platforms, we help you make informed decisions and progress your pipeline with confidence.
Our scientists specialize in the intricate challenges of ophthalmic drug development. We offer specialized solutions for:
- Targeted Compound Delivery: We are experts at administering compounds to specific ocular compartments, ensuring your drug’s pharmacology is tested in the most relevant biological context.
- Integrated Evaluation: Our platform combines disease models with a full suite of multimodal imaging and histology, providing comprehensive data on a drug’s effects on both the anterior and posterior segments of the eye.
- Expert Oversight: Every study is conducted to the highest scientific standards guided by experienced study directors, board-certified veterinary ophthalmologists, board-certified toxicologists, and in full compliance with animal welfare regulations, overseen by our in-house Institutional Animal Care and Use Committee (IACUC) and Institutional Biosafety Committee (IBC), experienced team of board-certified veterinary ophthalmologists, attending veterinarians, and skilled veterinary technicians.
Routes of Delivery:
- Topical
- Intrastromal
- Intracameral
- Intravitreal
- Pararetinal
- Subretinal
- Suprachoroidal
- Subconjunctival
- Subtenon
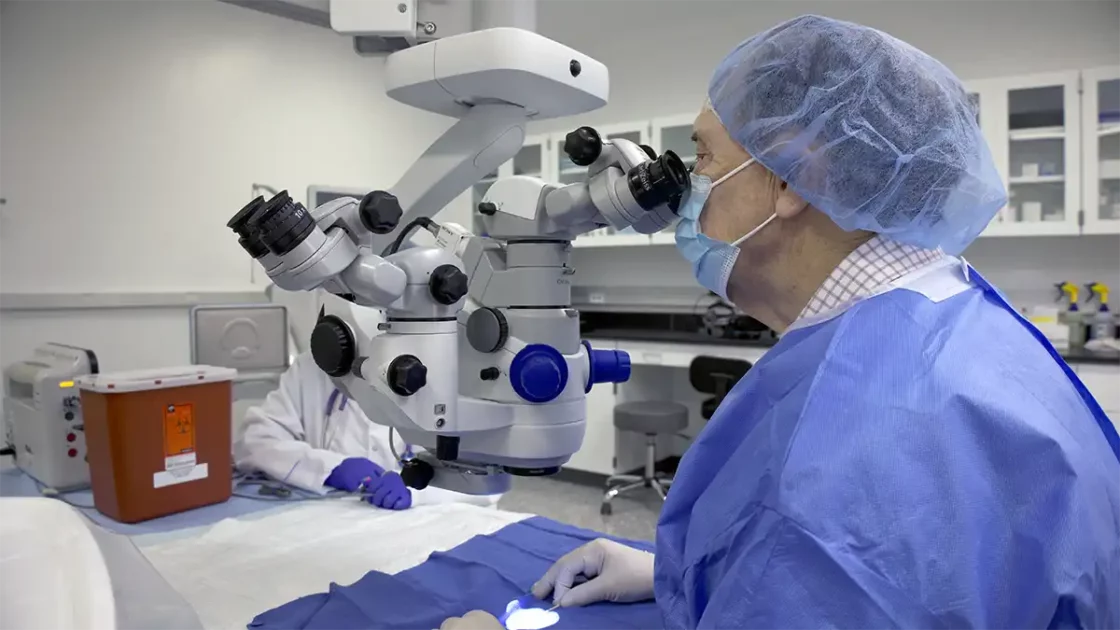
Imaging Modalities and Ocular Evaluation
- Surgical/operating microscope – videography
- Slit-lamp Photo??
- Pupillometry??
- Pachymetry??
- Specular Microscopy??
- Gonioscopy??
- Ultrasound Imaging?
- Fluorotron
- Color fundoscopy??
- Fundus autofluorescence??
- Fluorescein angiography??
Functional Assessment:
- Electroretinography
- Optokinetic response (rodents)??
- Visual evoked potential (VEP)?
Experimental Models


