
in vivo Pharmacology

in vivo Pharmacology
Testing the efficacy of a new compound in animals for the first time is an exciting moment in a drug discovery program. Partnering with Pharmaron ensures this critical phase is supported by access to over 1,850 in vivo disease models, expert teams, and state-of-the-art facilities to deliver reliable and reproducible results. Our facilities in China and the US offer a comprehensive array of resources that span a broad range of therapeutic areas.
A Holistic Approach to Meaningful Animal Testing
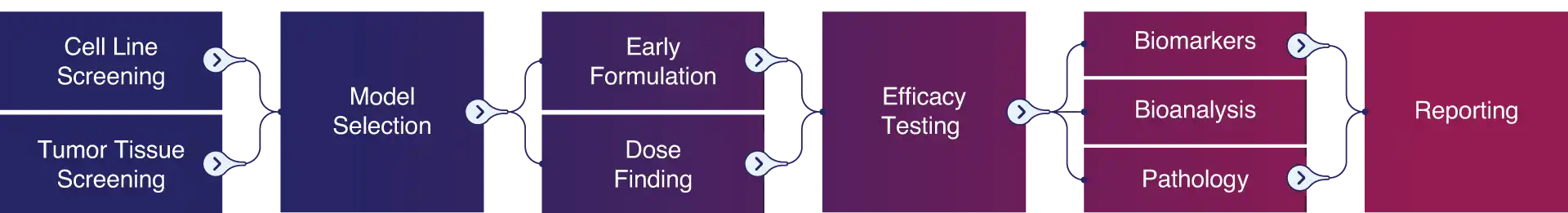
Pharmaron comprises experts covering every aspect of successful and meaningful in vivo testing. Our teams frequently develop new models to anticipate the needs of our clients. Formulation and toxicology experts pave the way for optimal efficacy testing. Our team provides state-of-the-art biomarker, bioanalysis, and pathology testing to ensure our clients get the most meaningful results possible.
Research Quality:
Clients consistently rate our in vivo pharmacology team as ‘excellent’ for fast turnaround, effective communication, and high-quality results. Our teams are strategically set up to enhance the client experience. For example, each client works with a dedicated study director for all study types, ensuring streamlined communication between the client and the executing teams.
AAALAC Accreditation:
Pharmaron’s compliance with international standards, including using EU standard cages for most studies, gives our clients confidence that our operations meet rigorous animal welfare and ethical research practices. Our in-house Institutional Animal Care and Use Committee (IACUC) and Department of Lab Animal Medicine (DLAM) oversee all animal welfare and care aspects.
Short Lead Times:
We work closely with our clients to develop customized study proposals, ensuring each study is tailored to their specific requirements for effective execution. After finalizing the study design, we promptly initiate animal procurement and other study-related preparations, enabling a swift study launch.
Animal Species:
We have the expertise and resources to conduct in vivo studies in many animal species, including transgenic animals. This approach allows us to cover a broader range of therapeutic areas, accommodating complex conditions. The diversity of our offerings enhances translational research, resulting in more reliable data.
We invite you to connect with us to explore how our dedicated in vivo pharmacology services team at Pharmaron can support your research endeavors.
Pharmaron’s in vivo Models by Therapeutic Area
More than 400 clients worldwide benefit from our comprehensive in vivo model offerings
Pharmaron’s comprehensive coverage of therapeutic areas and in vivo models allows us to conduct drug discovery programs tailored to specific research needs. This expertise helps address complex research questions efficiently, thereby accelerating research progress.

Our Experience Covers a Large Range of Modalities
At Pharmaron, our expertise spans a wide array of therapeutic modalities, enabling us to deliver tailored solutions and innovative approaches for efficacy testing potential new medicines.
- Small molecules
- Large molecules
- Antibodies
- ADCs and other conjugates
- Nucleic acid-based therapeutics
- LNPs as vectors
- Other biologics
- Cell and gene therapies
- Medical devices
- Radiotherapies
- Vaccines

ex vivo Pharmacology and Biomarkers
Pharmaron’s dedicated biomarker team quantifies the test article’s interaction with its target and the subsequent biological effects to enable the interpretation of efficacy study results.
- Analysis of body fluids and tissue samples
- Gene and genomics analysis
- Protein and proteomics analysis
- Flow cytometry and cell sorting
- Immunology analysis
- Histology, immunohistology, RNAscope
- Clinical pathology

PK/PD Studies
Our in vivo pharmacology team has bioanalysis expertise to measure the relationship between test article exposure, the time course of test article action, and the resulting therapeutic effects. In cooperation with our colleagues from the DMPK department, we utilize state-of-the-art modeling tools to predict therapeutic outcomes.
Related Resources:


Download PDF to learn more about Pharmaron’s in vivo Pharmacology Services


Learn how our in vitro biology and in vivo pharmacology teams work together

How can we support your animal testing project?


