
Cell and Gene Therapy (CGT) Ophthalmology Services

Cell and Gene Therapy (CGT) Ophthalmology Services
Pharmaron specializes in preclinical services for CGT ophthalmology products, helping you advance your therapies to First-in-Human (FIH) trials with confidence. We provide a comprehensive platform that combines reliable disease models with advanced, integrated evaluations. Our services are built to address the specific needs of CGT ophthalmology, featuring:
- Targeted Disease Models: Our in vivo models accurately mimic human ocular conditions for robust efficacy testing. We have deep expertise in the complex delivery of CGT ophthalmology products to both the front and back of the eye.
- Integrated Analysis: We combine multimodal imaging, histology, and bioanalytical assays to provide a complete picture of your product’s performance.
- Expert Oversight: Our team includes experienced Study Directors and support from board-certified veterinary ophthalmologists and surgeons, board-certified toxicologists, ensuring every study meets the highest standards of scientific rigor and regulatory compliance.
Partner with us to leverage our specialized expertise and accelerate your path to the clinic.
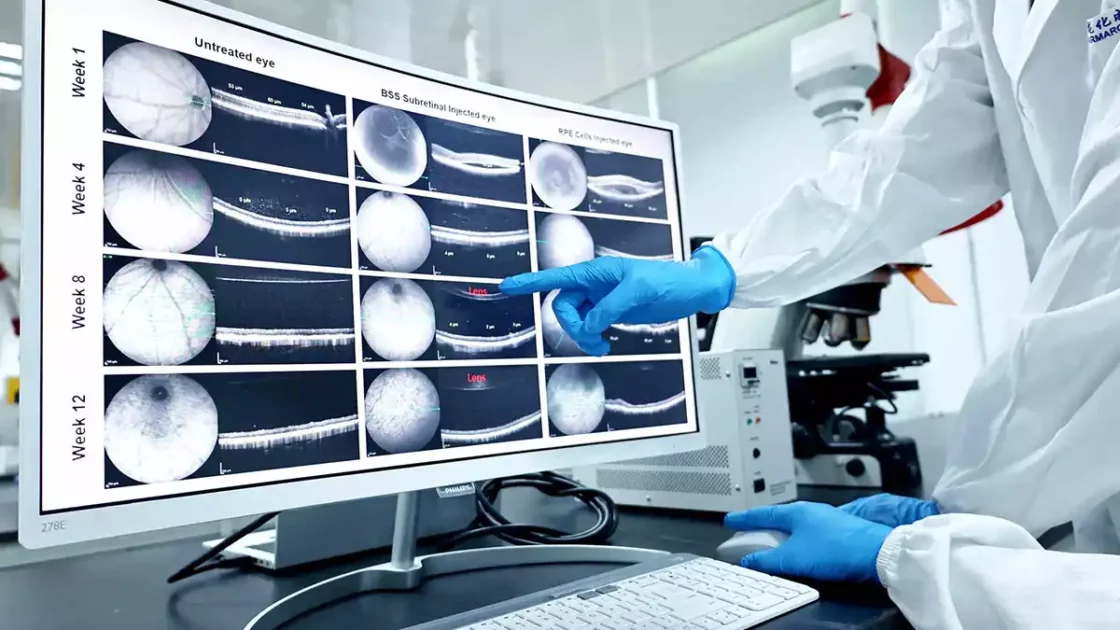
Supporting all services from compound administration through analytics
- Proof of Concept
- IND-enabling GLP Safety Evaluation
- In vivo Pharmacology Assessment
- Biodistribution
- Vector Shedding
- Immunogenicity Assessment
- Transgene and Protein Expression
- Potency Assays

Ocular Dose Routes for CGT Products
- Posterior Segment Targets (e.g., retina, RPE, choroid)
- Subretinal injection – Delivers therapy directly under the retina, often used for gene therapy targeting photoreceptors/RPE.
- Intravitreal injection – Into the vitreous humor; less invasive, used for inner retina delivery (e.g., ganglion cells), but limited for outer retina targets.
- Suprachoroidal injection – Between sclera and choroid; can target posterior tissues with less retinal detachment risk.
- Transscleral (sub-Tenon/episcleral) delivery – May be used for periocular sustained release or targeting choroid.
- Anterior Segment Targets (e.g., cornea, conjunctiva, trabecular meshwork)
- Intracameral injection – Into anterior chamber for aqueous humor–facing tissues.
- Intrastromal corneal injection – Directly into corneal stroma for corneal disorders.
- Topical application – Rare for CGT, but possible for certain gene therapy vectors if corneal epithelium is the target.
- Surgical Implantation Routes
- Epiretinal or subretinal scaffold/cell sheet implantation – For cell-based retinal replacement.
- Biodegradable or non-degradable ocular implants – Slow release of therapeutic cells or vectors.


