GTI Symposium 2025

Thank you for joining Pharmaron on September 18, 2025, at the GTI Symposium 2025
The Pharmaron team was proud to sponsor the Gene Therapy Initiative (GTI) Symposium 2025 at UC San Diego, taking place September 18 from 8:30 AM to 4:30 PM at the Conrad T. Prebys Auditorium, The Salk Institute, La Jolla, CA.
Join Manindra Singh, Associate Director of Business Development, Ophthalmology, Cell & Gene Therapy, and Natalie Fredette, Manager of Business Development for US Labs, Cell & Gene Therapy, who were excited to host a table and meet you at the GTI Symposium. We hope you found our table and connected with our team to learn how our expertise can help advance your gene therapy initiatives, but if you missed us – please use the form below to schedule a meeting so we can answer your questions.
Event Details:
- September 18, 2025
- Conrad T. Prebys Auditorium, The Salk Institute, La Jolla, CA
Connect with us
We would still love to connect with you to learn more about what you are doing and how we may be able to support or partner with you to expedite your path forward. Request a meeting by filling out the form below!
Our GTI Symposium Schedule
On-Site Hours
- September 18 from 8:30 am to 5:30 pm
- We will also be attending the Social Hour at 4:35pm following the poster and networking session and look forward to connecting with you there.
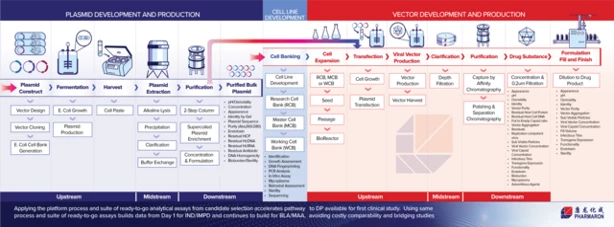
Our Cell & Gene Therapy Capabilities
Pharmaron offers comprehensive laboratory services designed to support the development of cell and gene therapy (CGT) products at every stage, from discovery and preclinical studies through clinical development and commercialization.
Our CGT laboratory technologies include:
- in vitro Assays – Development and validation of robust, reliable methods to support the analysis of CGT products
- in vivo Studies – Expertise in disease models for efficacy, safety, and pharmacokinetics
- Biodistribution and viral shedding – Advanced bioanalytical methods to assess vector presence and gene expression
- Cell and Gene Therapy (CGT) bioanalysis – Assay development to characterize CGT products and support regulatory submissions
- Cell and Gene Therapy (CGT) Ophthalmology Services – Ocular disease models and diverse dose routes for translational preclinical evaluation
- Cell and Gene Therapy (CGT) Safety Assessment – Addresses preclinical safety evaluation needs to support IND and BLA filings with regulatory agencies (FDA/EMA/NMPA)
Our modalities include medicinal products using AAV (various serotypes), viral and non-viral vectors, and cell-based therapeutics. We conduct studies under GLP, GCP, GCLP, and GMP guidelines, supporting IND and BLA submissions to FDA, EMA, NMPA, and other global regulatory agencies. Our capabilities also extend to clinical and commercial batch release, ensuring regulatory compliance and quality at every step.
If you’re attending, visit our table to meet with our team and discuss your upcoming projects to learn how Pharmaron can partner with you to accelerate your CGT programs. If you’re unable to attend, click the option on this page to schedule a meeting with Mani and Natalie to discuss your needs and learn how our flexible and integrated services can help bring your vision to reality.
About the Gene Therapy Initiative (GTI) Symposium 2025
The GTI Symposium brings together leaders from academia, industry, government, and patient advocacy to share scientific advances, foster collaborations, and drive the development of cell and gene therapies. Last year’s event hosted over 200 attendees and included presentations from UCSD, CIRM, NIH, the Cystinosis Research Foundation, and 23 poster presentations.? For more information and event registration, please visit the GTI Symposium website.
Request a Meeting
Meeting Request Form
Meeting requests during a conference or event