Home - About Us - Upcoming Events - 32nd Annual ESGCT 2025 Congress
32nd European Society of Gene & Cell Therapy Annual Congress


Join Pharmaron from October 7-10 at the Annual ESGCT Congress.
Pharmaron is delighted to attend and present at the 32nd European Society of Gene & Cell Therapy Annual Congress between October 7 – 10, 2025 in Seville, Spain.
We are available to discuss how we could be your global partner for your advanced therapy program, supporting your preclinical and clinical needs, your CMC/manufacturing, and your analytical testing.
Event Details:
- October 7 – 10, 2025
- Fibes Palacio de Exposiciones y Congresos, Avda. Alcalde Luis Uru?uela, 1 41020 Seville Spain
Connect with us
We would love to connect with you to learn more about what you are doing and how we may be able to support or partner with you to expedite your path forward. Find us during the conference, catch our poster, or request a meeting by filling out the form below!
ESGCT Annual Congress Schedule
On-Site Hours
- Tuesday, October 7: 8:30 am to 9:00 pm
- Wednesday, October 8: 8:00 am to 7:30 pm
- Thursday, October 9: 8:00 am to 7:30 pm
- Friday, October 10: 9:00 am to 6:00 pm
Posters
- Download Poster P0198 Here: Accelerated Analysis of Polysorbate 20 and Poloxamer 188 in AAV Samples using Mixed Mode UHPLC-ELSD
- Presented by Andrew Duckworth
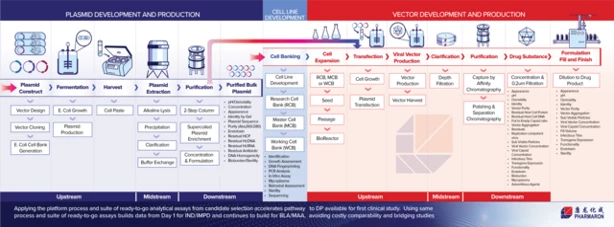
Our Cell & Gene Therapy Capabilities
Pharmaron offers comprehensive laboratory services designed to support the development of cell and gene therapy (CGT) products at every stage, from discovery and preclinical studies through clinical development and commercialization.
Our CGT laboratory technologies include:
- in vitro Assays – Development and validation of robust, reliable methods to support the analysis of CGT products
- in vivo Studies – Expertise in disease models for efficacy, safety, and pharmacokinetics
- Biodistribution and viral shedding – Advanced bioanalytical methods to assess vector presence and gene expression
- Cell and Gene Therapy (CGT) bioanalysis – Assay development to characterize CGT products and support regulatory submissions
- Cell and Gene Therapy (CGT) Ophthalmology Services – Ocular disease models and diverse dose routes for translational preclinical evaluation
- Cell and Gene Therapy (CGT) Safety Assessment – Addresses preclinical safety evaluation needs to support IND and BLA filings with regulatory agencies (FDA/EMA/NMPA)
Our modalities include medicinal products using AAV (various serotypes), viral and non-viral vectors, and cell-based therapeutics. We conduct studies under GLP, GCP, GCLP, and GMP guidelines, supporting IND and BLA submissions to FDA, EMA, NMPA, and other global regulatory agencies. Our capabilities also extend to clinical and commercial batch release, ensuring regulatory compliance and quality at every step.
If you’re attending, visit our table to meet with our team and discuss your upcoming projects to learn how Pharmaron can partner with you to accelerate your CGT programs. If you’re unable to attend, click the option on this page to schedule a meeting with us to discuss your needs and learn how our flexible and integrated services can help bring your vision to reality.
About the European Society of Gene & Cell Therapy Annual Congress
The European Society for Gene and Cell Therapy (ESGCT) is dedicated to fostering advancements in gene therapy, cell therapy, and genetic vaccines by facilitating education, promoting the exchange of information, and advising stakeholders in Europe.
For more information and event registration, please visit the ESGCT Congress website.
Request a Meeting
Meeting Request Form
Meeting requests during a conference or event